Metrology Basics
Training Guideline FDA 21 CFR Part 820, eLearning
600033-0201-905
- product type
- eLearning
- Training type
- eLearning
- Level
- 1
Overview about guideline 21 CFR Part 820
Code of Federal Regulation of Food and Drug Administration (FDA)
- purpose, scope, application area and general contents
- identification of specific requirement as per 21 CFR Part 820
- application of concepts for quality assurance
Duration: approx. 4 hours
Product note
ATTENTION! Contents of this eLearning only available in English language.
Recently Viewed
Training Guideline FDA 21 CFR Part 820, eLearning
600033-0201-905
149,00 €excl. VAT
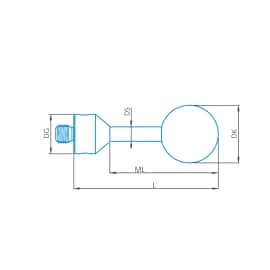
Stylus straight M5, DK12 L25
626115-1200-025
213,00 €excl. VAT
Available
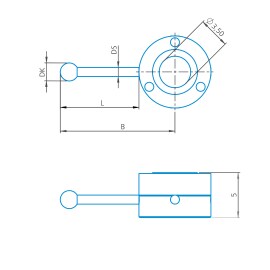
L-stylus cube M3 XXT, DK1.5 L5
626103-0150-418
147,00 €excl. VAT
Available
Stylus straight M5, DK8 L63.5
612030-9649-000
103,00 €excl. VAT
Available
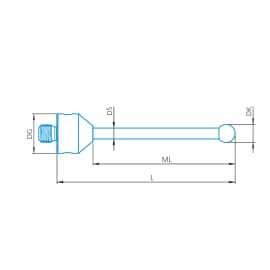
Stylus straight M5, DK5.5 L30
626115-0551-030
71,80 €excl. VAT
Longer delivery time
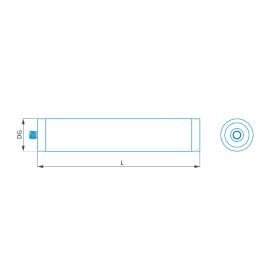
Cylinder stylus M5, DK5 L40
626115-0501-040
55,00 €excl. VAT
Made to Order